Pharmaceutical Solutions
Pharmaceutical operational excellence requires more than just meeting standards; it needs integrated data systems that bridge the gap between research, manufacturing, and distribution while ensuring absolute compliance and data integrity.

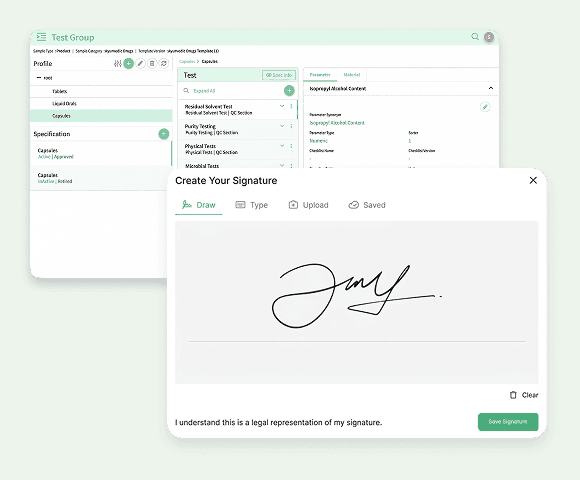
LIMS
A Laboratory Information Management System (LIMS) is the operational core of regulated pharma or biotech labs. It tracks samples from receipt to result, links results to relevant instruments, and maintains audit-ready records.
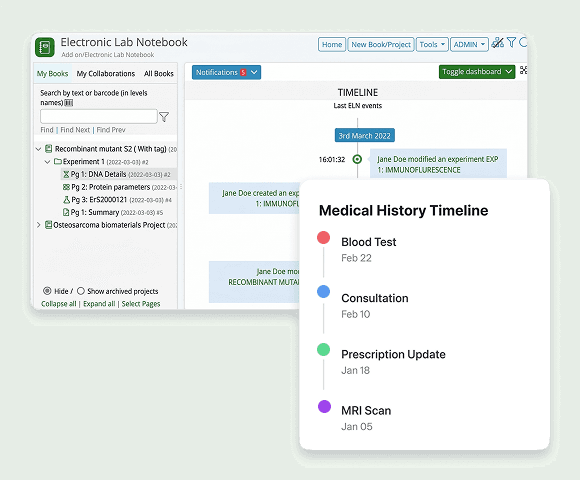
Electronic Lab Notebook
Paper lab notebooks create data integrity risks. Our Electronic Lab Notebook (ELN) ensures structured data capture, contemporaneous recording, and 21 CFR Part 11 compliant electronic signatures for research and development.
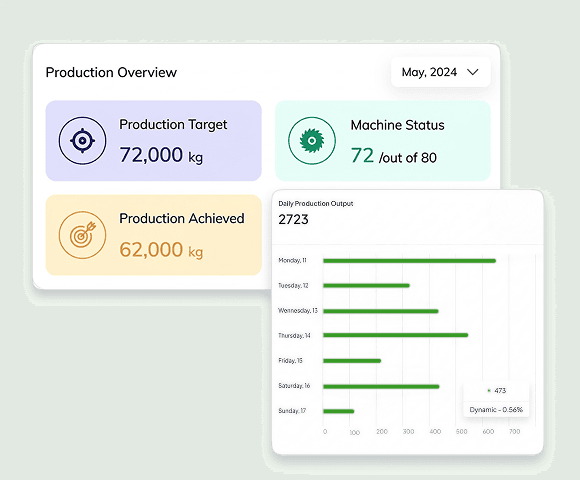
MES (Manufacturing Execution System)
Manufacturing Execution Systems link ERP planning with the production floor. Our MES solutions enable paperless batch record execution, real-time operator confirmations, and critical process parameter monitoring.
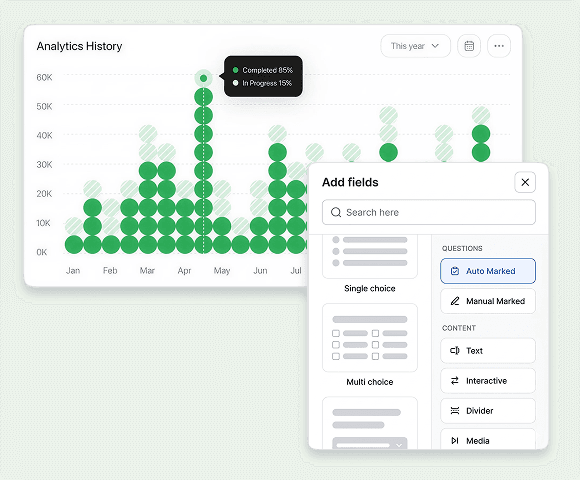
Compliance Management
Pharma quality compliance is a connected system where deviations lead to investigations, CAPAs, and changes. Our systems provide a closed-loop quality audit standard matching FDA and EMA requirements.
Clinical Trial Management
Clinical trial management requires tracking site activation, monitoring enrollment in real-time, and managing protocol deviations. Our CTMS ensures data-intensive trials are managed according to GCP guidelines.
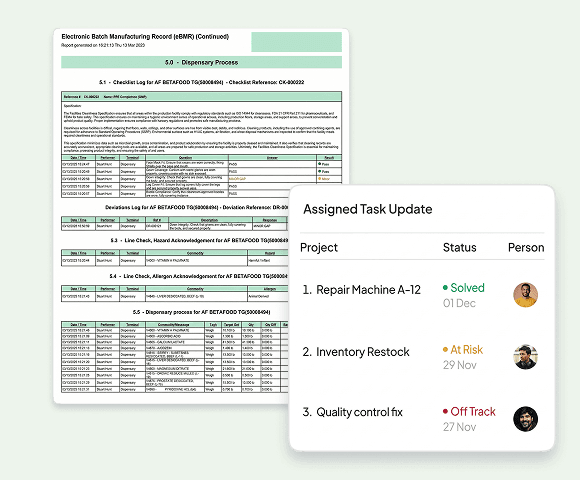
Electronic Batch Record Management
Batch records are critical in pharma manufacturing. Our EBR systems prevent out-of-sequence work, enforce mandatory in-process controls, and provide tamper-evident audit trails for 21 CFR Part 11 compliance.
